Whatcom sees 13 new COVID-19 cases, state reports Wednesday; CDC redefines ‘close contact’
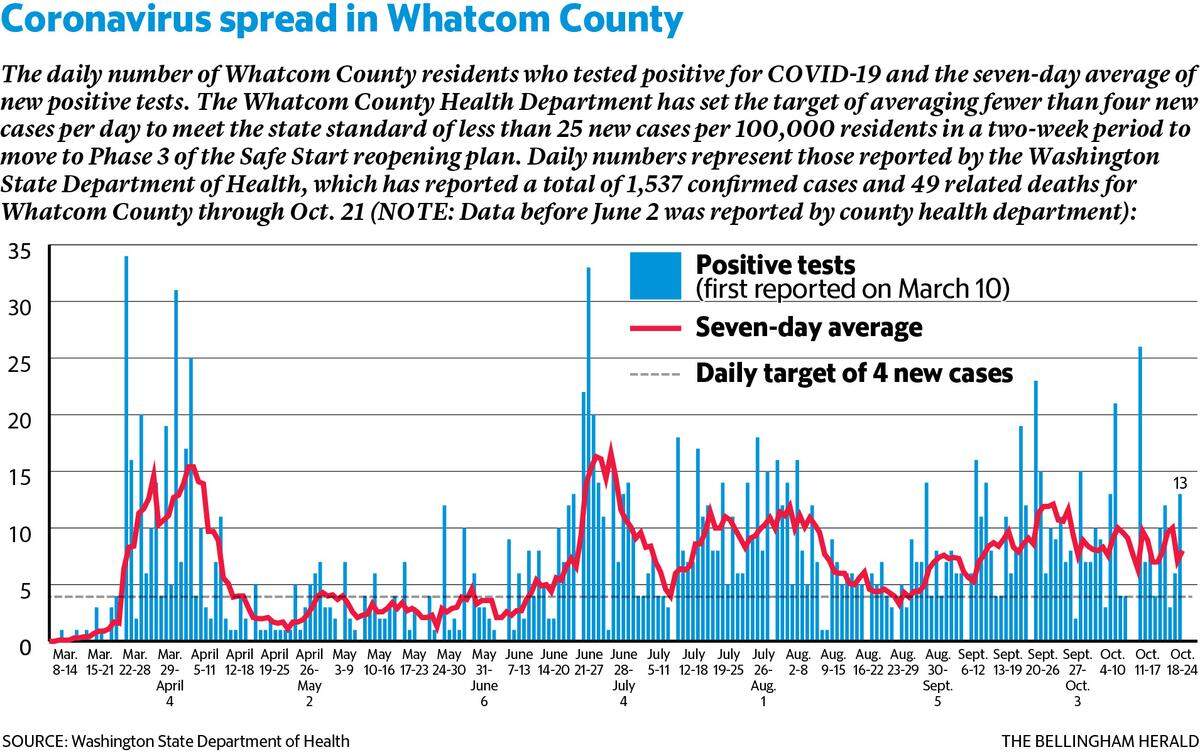
Thirteen more Whatcom County residents have tested positive for COVID-19, the Washington State Department of Health reported Wednesday, Oct. 21, but no new deaths were reported.
The county now has seen 1,537 confirmed cases and 49 related deaths during the pandemic, according to state data as of 11:59 p.m. Tuesday, Oct. 20. That means that 3.2% of the Whatcom residents who have tested positive for COVID during the pandemic have died.
The state Department of Health data also shows Whatcom County has had 106 hospitalizations (an increase of one from Tuesday) and 75,465 tests have been performed (an increase of 719 from Tuesday).
The state’s Risk Assessment Dashboard, updated Wednesday for data as of 11:59 p.m. Tuesday, showed that Whatcom County is making two of four Phase 2 metrics goals.
▪ Whatcom is missing the target rate of fewer than 25 new cases per 100,000 residents every 14 days with a rate of 51.5.
▪ Whatcom had an average COVID-19 testing rate per 100,000 people over a week of 245.8. No goal was stated for this metric, however, the overall statewide number was 237.3.
▪ Whatcom is making the target of less than 2% of individuals testing positive for COVID-19 during the past week with a rate of 1.7%.
▪ Whatcom is missing the target of less than 80% of all licensed hospital beds occupied by patients with 83.5% reported by the state.
▪ Whatcom is making the target of less than 10% of all licensed hospital beds being occupied by COVID-19 patients with 1.6% reported by the state.
St. Joseph hospital in Bellingham reported to The Bellingham Herald on Wednesday that it was treating four patients for COVID-19, a number unchanged from Tuesday.
Western Washington University’s most recent on-campus student COVID testing data shows that, since Sept. 15, WWU has tested 4,861 students, five of whom had positive tests.
Numbers elsewhere
COVID-19 cases and deaths according to Johns Hopkins University Wednesday afternoon:
▪ The U.S. has more than 8.3 million reported cases, the most of any nation, and 221,862 deaths.
▪ Worldwide, there are more than 41 million reported cases and 1.1 million deaths.
In Washington state, the most recent numbers from the Department of Health were reported Wednesday afternoon:
▪ 99,874 reported cases, an increase of 724 from data on Tuesday.
▪ 8,149 coronavirus-related hospitalizations, an increase of 25 from data Tuesday.
▪ 2,276,453 total tests, an increase of 23,665 from data Tuesday.
▪ 2,286 deaths related to COVID-19, an increase of four from Tuesday — meaning that 2.3% of the state residents who have tested positive for COVID during the pandemic have died.
State vaccination plan
The Washington State Department of Health submitted its COVID-19 vaccination plan to the Centers for Disease Control and Prevention by the Oct. 16 deadline, according to a release Wednesday. The plan outlines the department’s preliminary distribution plans once the U.S. Food and Drug Administration approves a vaccine as safe and effective.
“We want to emphasize that this is the first version of our plan,” Acting Assistant Secretary with executive oversight for COVID-19 vaccines Michele Roberts said in the release.
“This is essentially a living document. But the one thing that won’t change is our focus on equitable distribution of the vaccine as a priority. As we learn more about the vaccine, and as we learn more specifically from communities and partners most impacted by COVID-19, this plan will evolve.”
Part of Washington’s plan says the state Department of Health will identify and train vaccine providers, and monitor data about vaccine administration.
They’ll also “gather feedback from communities that are disproportionately affected by COVID-19 or at higher risk of contracting the disease,” and will “provide timely, accurate, and credible information to the people of Washington on COVID-19 vaccine, including safety information,” the plan says.
The document talks about the different phases of vaccine distribution.
“Phase 1 vaccination will be at specific sites highly targeted at those recommended first to receive FDA approved vaccine that is safe and effective,” the plan says. “Recommendations for who receives vaccine in phase 1 will be based on ACIP (Advisory Committee on Immunization Practices) recommendations, the National Academies’ Framework for Equitable Allocation, and state allocation framework developed with input from partners and communities collected through mixed methods during fall 2020.”
Possible groups to get the vaccine in phase 1 may be “high-risk workers in health care settings,” “high-risk first responders,” “people of all ages with comorbidities,” “older adults in congregate/crowded settings,” and “essential workers,” the plan says.
In phase 2, it says: “When there is sufficient supply to meet demand, the state will need many vaccine administration locations. We will use a broad network of provider settings, including community health centers, pharmacies, primary care providers, community or business points of dispensing (PODs), long-term care facilities, congregate living facilities, and occupational health clinics. Both traditional and nontraditional vaccination sites will deliver vaccine to ensure that all people who are recommended to receive it have many access points.”
Next, the plans says: “Phase 3 moves to a steady state where there is sufficient supply to meet demand and vaccination continues to grow using routine provider networks proven to reach critical populations. While the department and our partners will promote completion of the vaccination series, phase 3 will be an opportunity to enhance efforts to remind or recall individuals to complete any missing doses.”
Vaccine trial volunteer dies
A Brazilian volunteer in the trial of a COVID-19 vaccine developed by AstraZeneca and Oxford University has died, according to a report by Reuters. The death was announced Wednesday by Brazilian health authority Anvisa, but the trial is expected to continue.
Citing Brazillian press reports, The Wall Street Journal reported that the volunteer was a man in his 20s from Rio de Janeiro, but it was not known if he had received the vaccine or a placebo during the trial.
Last week, the Lummi Indian Business Council announced that it was withdrawing from the AstraZeneca trial. The primary reason for the decision, according to a release, was ongoing communications problems between Lummi doctors and AstraZeneca representatives.
“We will continue to look for ways to protect our people from this virus,” Lummi Indian Business Council Chairman Lawrence Solomon said in the release. “But after consultation with the Lummi Public Health Department, it was clear that the AstraZeneca vaccine trial was not a good fit.”
CDC redefines ‘close contact’
U.S. health officials Wednesday redefined what counts as close contact with someone with COVID-19 to include briefer but repeated encounters.
For months, the Centers for Disease Control and Prevention said close contact meant spending a solid 15 minutes within 6 feet of someone who tested positive for coronavirus. On Wednesday, the CDC changed it to a total of 15 minutes or more — so shorter but repeated contacts that add up to 15 minutes over a 24-hour period now count.
The CDC advises anyone who has been in close contact with a COVID-19 patient to quarantine for two weeks.
The change may prompt health departments to do contact tracing in cases where an exposure might previously have been considered too brief, said Dr. William Schaffner, a Vanderbilt University infectious diseases expert.
It’s also serves notice that the coronavirus can spread more easily than many people realize, he added.
Phased reopening
Gov. Inslee on Tuesday, Oct. 13, moved all counties in modified Phase 1 to Phase 2, but his July 28 extension of an indefinite pause on counties moving ahead in the Safe Start Washington plan remains in place. That came a week after Inslee loosened some restrictions for activities and businesses.
That means 22 counties — including Whatcom — are in Phase 2 and 17 counties are in Phase 3.
Alexis Krell of the Tacoma News Tribune and the Associated Press contributed to this story.
